Enhancing the way immune cells respond to a threat
VIB researchers uncover a metabolic switch that allows immune cells to enhance their protective function.
Immune cells protect our body against external (bacteria, viruses) and internal (cancer) threats. Among these cells, cytotoxic— or killer—T cells can rapidly activate to attack the infected or cancerous cells. They also acquire a long-term memory that helps them fight recurring infections or tumors. In each of these different stages, T cells rely on different nutrients—in other words, their metabolism changes. The team of Prof. Sarah-Maria Fendt (VIB-KU Leuven Center for Cancer Biology) identified a metabolic switch that allows these T cells to enhance their protective function. Their work appears in Cell Reports.
In short:
- To respond to bacteria, viruses, or tumors, killer T cells must differentiate to an 'effector' state that allows them to attack the threat.
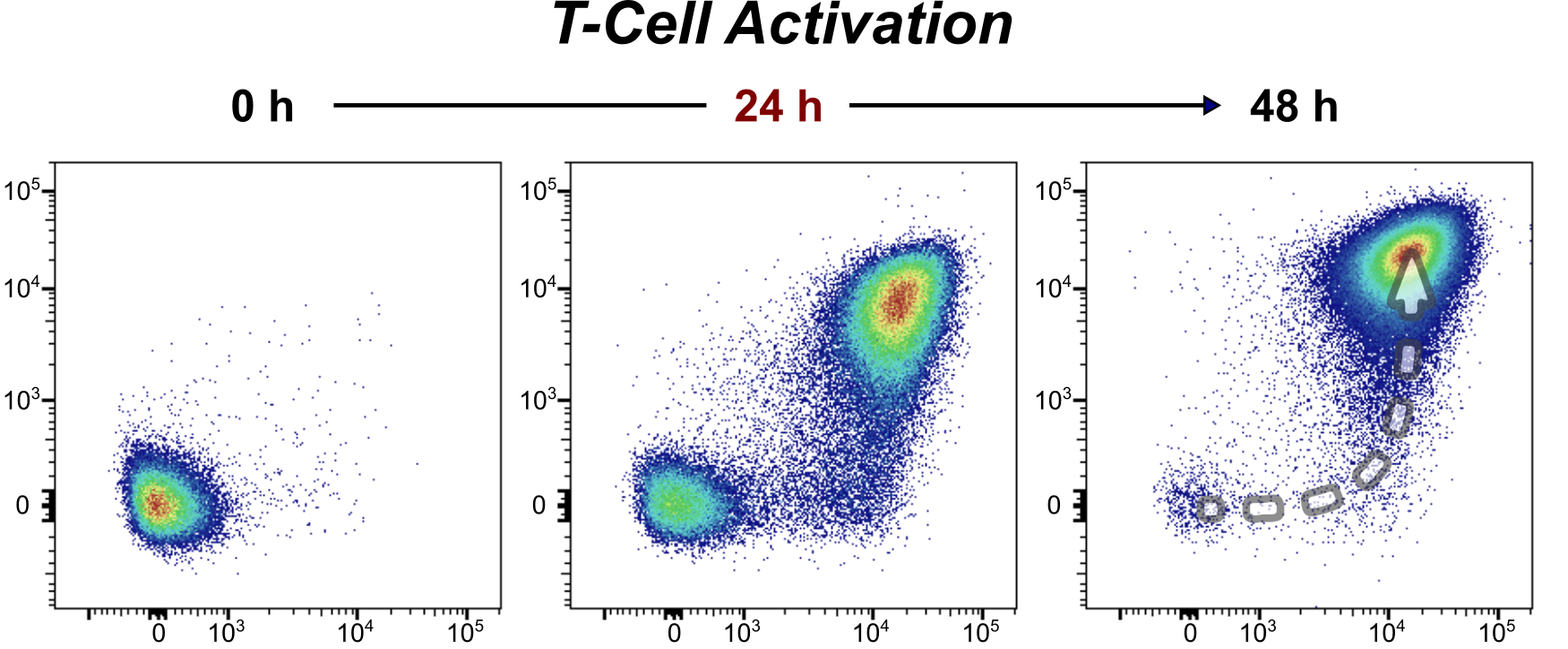
- Single-cell RNA sequencing shows that the metabolic protein asparagine synthetase (ASNS) is crucial for attaining this effector state.
- Overexpressing ASNS in mouse T cells increased their ability to respond to viral infection and fight skin cancer.
The link between T cell metabolism and function
Cytotoxic T cells play the role of soldiers in our immune system. Like all soldiers, they require training, deployment, and nutritious food. The process by which these cells take up nutrients from their environment and use them to support their function is known as cellular metabolism. However, the details of how cellular metabolism supports T-cell responses are not entirely known. Scientists have some good clues, though. For example, as T cells evolve through different states during an immune response, they will use different nutrients. This is called metabolic rewiring—the nutritional needs are not the same for resting, effector, and memory T cells.
Prof. Sarah-Maria Fendt (VIB-KU Leuven Center for Cancer Biology): "Despite our knowledge of T-cell metabolism at different functional states, little is known about how it dynamically evolves when transitioning between them. With single-cell RNA-sequencing, we can now provide a global description of this dynamic metabolic rewiring, and use it to define yet unknown metabolic dependencies of T cells."
With single-cell RNA sequencing, Prof. Sarah-Maria Fendt's team investigated gene activity at the single-cell level. They identified how different metabolic genes dynamically evolve during T-cell responses. A metabolic requirement for an effective immune response is the expression of the metabolic protein asparagine synthetase (ASNS).

Metabolically boosting T-cell fitness
To see whether ASNS actually plays a role in living organisms, the researchers overexpressed the protein in T cells from donor mice. Recipient mice injected with T cells with higher ASNS expression were better able to respond to a viral infection, and they also showed a better anti-tumor response when dealing with melanoma.
Dr. Juan Fernández-García, first author of the study: "Increasing ASNS expression in T cells enhanced the ability of the mice to respond to a viral infection and to clear melanoma tumors. This suggests that ASNS is an interesting protein to be overexpressed in CAR-T cells for cancer immunotherapy. Importantly, our work provides a global resource that can be used by other researchers to identify new metabolic targets to improve T-cell fitness."
Publication
CD8+ T Cell Metabolic Rewiring Defined by scRNA-Seq Identifies a Critical Role of ASNS Expression Dynamics in T Cell Differentiation. Fernández-García et al. Cell Reports 41, 111639, 2022.