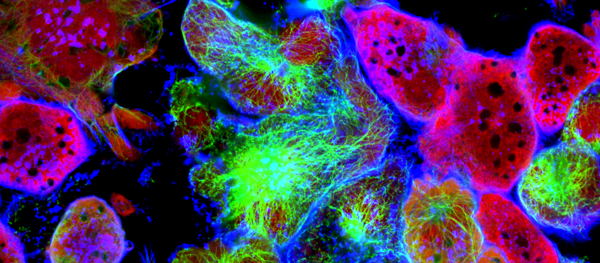
How autophagy shapes the lymphatic system
Our lymphatic vasculature is part of our circulatory system and it carries 'lymph' through our body. This is crucial to maintain internal fluid levels, transport immune cells to their location, and for fat absorption. New work by the team of Prof. Patrizia Agostinis (VIB-KU Leuven Center for Cancer Biology and the Department of Cellular and Molecular Medicine at KU Leuven) shows how autophagy shapes our lymphatic system. Their results appear in the journal Nature Communications.
Facts about the lymphatic system
- Starts developing around the fifth week of embryonic development
- In adults, problems with the lymphatic vessels can lead to fluid retention, which manifests as tissue swelling (lymphedema) and impaired wound healing
- The excessive formation of new lymphatic vessels (called lymphangiogenesis) can favor the spread of cancer
- The lymph nodes in our body serve as battlefields for our immune system to remove toxins, waste, and pathogens
- An increasing number of human diseases are linked to dysfunctions of the lymphatic system
The importance of autophagy
Autophagy (literally 'self-eating') is the major mechanism for getting rid of damaged or old cell materials by taking them to the lysosomes. These lysosomes are cellular structures in which damaged cell components are broken down and recycled as a source of energy. This is good for us because it helps our body get rid of damaged cellular components and pathogens, and it makes sure that all of our cells and tissues function well, especially when nutrients are scarce.
The team of Prof. Patrizia Agostinis (VIB-KU Leuven Center for Cancer Biology and the Department of Cellular and Molecular Medicine at KU Leuven) now demonstrates that autophagy is also vital for the lymphatic system. Using both human cell lines and lab mice, the researchers show that autophagy is a critical process for the growth of the lymphatic vessels in response to injury-causing inflammation. Losing the capacity for autophagy in the lymphatic vessels impairs their ability to respond to growth-stimulating factors.
Fatty acids and mitochondria
Agostinis: "It was clear that autophagy had a large effect on wound-healing and inflammation, but we wanted to discover how this happened. Because lymphatic vessels are specialized in absorbing and moving around fatty acids, and they ‘eat’ fat as nutrients for their growth, that's where we started."
It was a good call.
The team found that autophagy in the lymphatic endothelial cells – the building blocks of the lymphatic vessels – controls the breakdown and recycling of specific vesicles, called lipid droplets, where fats are stored within the cell before they can be used as nutrients in metabolism. A selective autophagy process – termed lipophagy – then cuts up these vesicles and frees the nutrients for the powerhouses of our cells, the mitochondria. Once delivered to the mitochondria, the fatty acids are used to produce energy and much more.

The use of these nutrients by the mitochondria also generates molecules that instruct the nucleus of the cells to keep up the expression levels of VEGFR3. This is a crucial receptor that, when stimulated by the protein VEGF-C, tells the lymphatics to proliferate and migrate to form new vessels. When autophagy in the lymphatic endothelial cells does not work, the fatty acids don't make it to the mitochondria. Result? The lymphatic vessels can’t eat their fatty meal and do not grow in response to a traumatic injury.
Dr. Odeta Meçe, co-first author of the study: "We also show that supplementing mice with acetate, a fatty acid precursor that the mitochondria can directly use, restores the growth of lymphatic vessels - even when autophagy is not functioning well.”
Diede Houbaert, PhD student and co-first author, adds: "Our findings further emphasize the vital connection between mitochondria fatty acid metabolism and lymphatic vessels growth recently highlighted by the group of Prof. Peter Carmeliet.”
Agostinis “The link between autophagy and lymphatics is intriguing also considering that a high-fat, low carbohydrate ketogenic diet and physical exercise alleviate lymphedema by stimulating the formation of lymphatic vessels are well-known stimulators of autophagy. Future studies are needed to better understand whether and how pharmacological modulation of autophagy might normalize dysfunctional lymphatic vessels in lymphedema and other critical disease conditions."
Meçe, Houbaert, et al. Nature Communications 2022
Questions from patients
A breakthrough in research is not the same as a breakthrough in medicine. The realizations of VIB researchers can form the basis of new therapies, but the development path still takes years. This can raise a lot of questions. That is why we ask you to please refer questions in your report or article to the email address that VIB makes available for this purpose: patienteninfo@vib.be. Everyone can submit questions concerning this and other medically-oriented research directly to VIB via this address.
Gunnar De Winter
.tif)