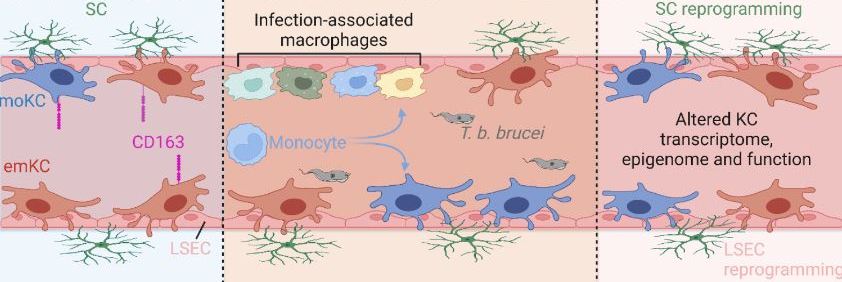
Past infections have long-term effects on liver immune cells
Many infections affect the liver, yet little is known about the long-term impact of these infections on the liver’s immune cells. Now, a study by the teams of Prof. Jo Van Ginderachter (VIB-VUB) and Prof Kiavash Movahedi at VUB reveals long-lasting changes in the liver’s Kupffer cells following an infection. The work appears in the Journal of Hepatology.
The role of Kupffer cells
Our immune system is a complex network of cells and molecules that work together to protect our bodies from harmful invaders like bacteria, viruses, and parasites. Among the key players in this defense system are macrophages, specialized immune cells that act as the first line of defense against infections. In the liver, a vital organ responsible for filtering, a specific type of macrophage called Kupffer cells plays a crucial role in monitoring the blood for pathogens.
Yet, the long-term effects of infections on the liver immune cells, including Kupffer cells, are unknown. Researchers from the lab of Prof. Jo Van Ginderachter at the VIB Center for Inflammation Research and Prof Kiavash Movahedi at VUB, with colleagues from UGent and the VIB Single Cell Core studied the lingering effects of infection with the parasite Trypanosoma brucei brucei on the liver immune cells (in a mouse model for human sleeping sickness).
.png)
A lasting mark
By using cutting-edge techniques such as fate mapping, single-cell sequencing, and epigenetic analysis, the researchers – led by PhD student Mohamed Amer Musrati and Dr. Benoit Stijlemans – acquired a detailed look at the changes in the liver following infection.
They found that Trypanosoma infections strongly alter the composition of macrophages in the liver, identifying several newly recruited populations that altered their gene expression to fight the infection. Remarkably, after the infection is cleared, the Kupffer Cells bear long-lasting changes in the accessibility and expression of certain genes, highlighting the enduring impact of past infections on our immune system.
Importantly, the mice who had gone through an earlier infection were more resilient to a subsequent infection with another pathogen, which suggests that an infection reprograms the liver’s Kupffer cells to better fight future infections. In other words, our infection history may influence the way we can deal with future infections due to macrophage reprogramming.
Prof. Jo Van Ginderachter says, “By unraveling the long-term effects of infections on liver macrophages, this study opens up new avenues for understanding and treating infectious diseases.”

Funding
This work was supported by FWO, ERC, an iBOF research consortium, and the European Regional Development Fund (ERDF).
Publication
Infection history imprints prolonged changes to the epigenome, transcriptome, and function of Kupffer Cells. Musrati, Stijlemans, et al. Journal of Hepatology, 2024.
Gunnar De Winter