Researchers discover cause of immunosuppressive behavior of regulatory T cells in cancer
Fucosylation – the addition of a fucose sugar to a molecule – has been identified as the driving force behind unbalanced Treg behavior in tumor microenvironment
Leuven, 13 November 2023 — A research team led by Professor Massimiliano Mazzone at the VIB-KU Leuven Center for Cancer Biology discovered a biological process responsible for unbalancing regulatory T cells (Tregs) in cancer. A research paper published in Cancer Immunology Research, details how the researchers found a metabolic switch that turns down the immune response of T cells against cancer cells, leading to a worse prognosis. The team also succeeded in preventing this process, increasing the effectiveness of the patient’s own immune system against cancer cells.
Regulatory T cells, or Tregs, are an essential part of the human immune system. In medicine, Tregs are considered to be the guardians of controlled immune reactions as they help prevent the development of autoimmune diseases. In cancer, however, the opposite is true. When confronted with tumors, Tregs proliferate at an unusually high rate, which prevents the immune system from destroying cancer cells. In doing so, Tregs can contribute to a worse disease outlook for patients. Researchers have so far been unable to identify the exact cause of this process. Until now.
Recent research already suggested that a protein called Foxp3, responsible for regulating the health and suppressive function of Tregs, could be linked to Tregs becoming immunosuppressive in the context of cancer. More specifically, metabolic changes in Tregs negatively affect Foxp3 levels. For instance, when Tregs are confronted with a tumor, the ability of cancer cells to alter their metabolism in support of their own growth can cause Tregs to be metabolically reprogrammed. In turn, researchers believe this reprogramming affects the immunosuppressive qualities of Tregs.
Rehabilitating Tregs
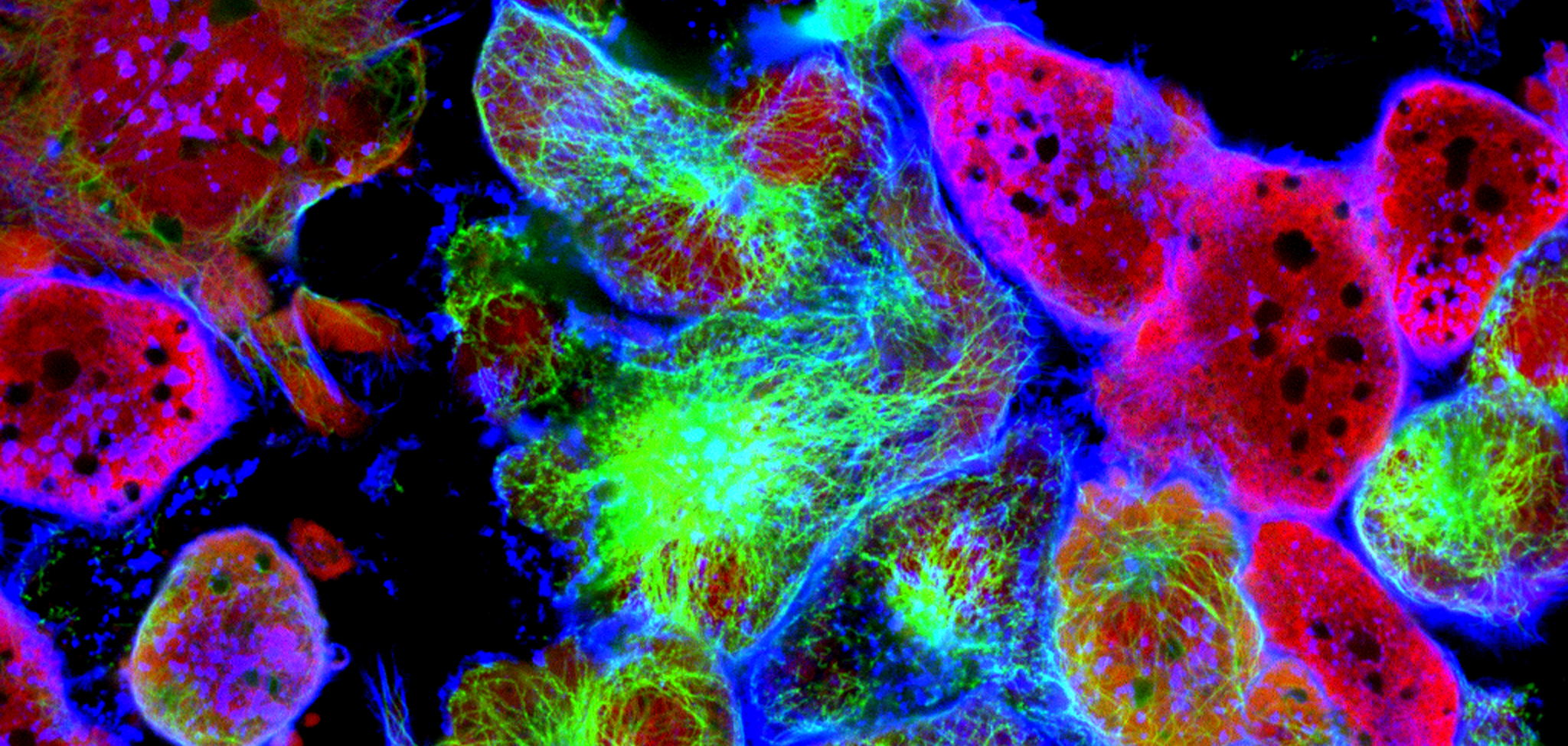
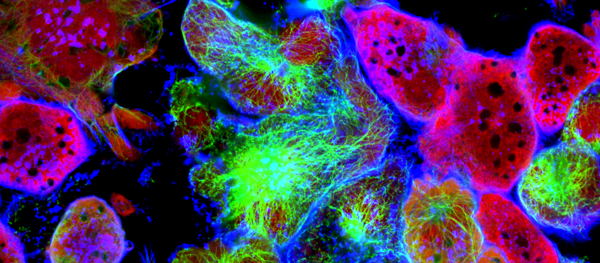
To confirm if this is indeed the case, the team of Prof. Massimiliano Mazzone (VIB-KU Leuven Center for Cancer Biology) employed CRISPR-Cas9 screening techniques to identify the underlying processes that drive the changes in Tregs behavior. The study revealed that fucosylation, a biological process in which so-called fucose sugar units are added to a molecule, is important for the proliferation and suppressive function of Tregs. These results were applicable both in vitro and in vivo.
Following this discovery, the team decided to test if halting fucosylation would diminish the immunosuppressive character of Tregs in the cancer microenvironment. And successfully so: by targeting the gene responsible for transporting fucose sugar units, the team was able to interrupt the fucosylation process in mice. This halted Treg functionality, which, in turn, normalized the tumor microenvironment and impaired tumor growth.
Professor Massimiliano Mazzone: “As Tregs play a major role in the prognosis of cancer and the development of autoimmune diseases, we decided to screen for metabolic regulators of Foxp3. Our findings show that fucosylation plays an important role in the tumor microenvironment and Tregs biology. We discovered that by tackling this biological process, we can reduce the immunosuppressive function of tumor Tregs, allowing the immune system to destroy cancer cells.”

From theory to practice
As a next step, the research team decided to investigate if their findings would also apply in humans. To do so, they analyzed tumor Tregs from colorectal cancer patients and clustered them together based on the levels of fucosylation. The team found that tumors infiltrated by Tregs with low levels of fucosylation-related genes had a better outcome and a greater response to immunotherapy, compared to those tumors characterized by Tregs with high expression levels of fucosylation-related genes.
Sotiria Pinioti, first author of the study and postdoctoral researcher at the VIB-KU Leuven Center for Cancer Biology: “Measuring the fucosylation levels in Tregs might pave the way towards a useful biomarker model for cancer patients. Other metabolic targets revealed from our screening, can also be used to enhance or inhibit Tregs function depending on the disease.”

The results of this study could ultimately lead to improved diagnostics tools for specific cancer types. However, more research will be needed to further explore the applications of this research.
Joran Lauwers
Questions from patients
A breakthrough in research is not the same as a breakthrough in medicine. The realizations of VIB researchers can form the basis of new therapies, but the development path still takes years. This can raise a lot of questions. That is why we ask you to please refer questions in your report or article to the email address that VIB makes available for this purpose: patienteninfo@vib.be. Everyone can submit questions concerning this and other medically-oriented research directly to VIB via this address.
About the VIB-KU Leuven Center for Cancer Biology
Cancer has many causes. Often it is a combination of lifestyle, environmental factors and genetic variation. We need to fight cancer on many fronts, and this can only be done by using knowledge. The VIB-KU Leuven Center for Cancer Biology researchers unravel new mechanisms in order to develop both specific diagnostic methods and treatments.