Understanding the role of microglia in Alzheimer’s disease
Leuven, Antwerp, and London, 27 March – Microglia are specialized immune cells in the brain. While they normally protect our brains, they can also contribute to neurodegenerative diseases such as Alzheimer's. The exact mechanism behind this contribution is not yet fully understood due to the complexities involved in studying them in human brain samples. Now, a research team led by Prof. Bart De Strooper (UK-DRI@UCL and VIB-KU Leuven) and Prof. Renzo Mancuso (VIB-UAntwerp) made a xenotransplantation model – mice with stem-cell-derived human microglia in their brains to observe how human microglia respond to the disease environment. Their findings, published in Nature Neuroscience, will help scientists better understand the complex mechanisms involved in Alzheimer's disease.
Alzheimer's disease (AD) is a complex, progressive neurodegenerative disorder that affects millions of people worldwide. The World Health Organization predicts a tri pling of cases by 2050, highlighting the urgent need for new treatments.
Microglia, our brain’s immune cells, are responsible for clearing debris and responding to inflammation in the brain. Scientists have been studying these cells in AD, as they play a central role in the disease, especially in the building-up of and early response to amyloid-β plaques, a hallmark of the disease. The microglia react to the plaques as they are perceived as foreign to the brain, making them the main drivers of the neuroinflammation that characterizes AD. Studying these cells in human brain samples post-mortem can be challenging because of genetic differences between people, the time between death and examination, and the presence of other brain disorders. Indeed, studies in human postmortem brain samples have shown mixed results regarding the reaction of microglia. It’s also not possible to test the effects of medication on post-mortem brains.
That is why the first authors of the study, Dr. Nicola Fattorelli and Dr. Anna Martinez Muriana, together with their colleagues at the VIB-KU Leuven Center for Brain & Disease Research, the VIB-UAntwerp Center for Molecular Neurology and the UK Dementia Research Institute, developed a unique mouse model. This xenotransplantation model is genetically engineered to mimic the amyloid-β plaque accumulations seen in humans with AD and can be transplanted stem-cell-derived human microglia. Previously, a similar model was able to show how transplanted human neurons die in AD. Now, this approach allowed the researchers to investigate how human microglia respond to amyloid plaques during the course of the disease.
Microglial responses to AD
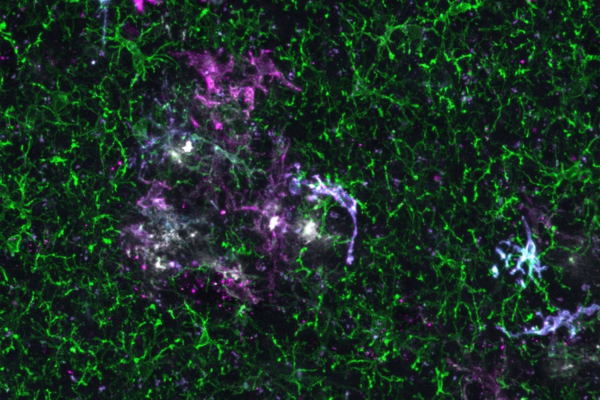
The scientists found that human microglia showed a much more complex immune response to amyloid-β than their rodent counterparts. Human microglia also displayed a different genetic transition from the normal to the reactive state of the cells.
“This could have implications for the development of treatments,” says Professor Renzo Mancuso, first author of the study and group leader at the VIB-UAntwerp Center for Molecular Neurology. “Researchers need to be cautious when using mouse models to study AD in preclinical systems for potential therapeutic targeting of microglia because the responses of human and mouse microglia may not be the same.”
Genetics and early intervention
The study also revealed that different genetic risk factors for AD influence how human microglia respond to the disease. Moreover, the genetic risk of AD was spread over the different reactive states of the microglia, further demonstrating the importance of microglia in the disease process. This suggests that future microglia-targeted therapies need to be implemented with care as genetic factors might differentially affect their cell states and modify the disease course in unpredictable ways.
Furthermore, the data hinted at a possible interaction between microglia and soluble forms of amyloid-β, which appear early in the disease process, well before plaques form. This interaction might occur in the very early stages of Alzheimer's and could potentially influence how the disease progresses. The question remains as to whether this microglial response affects neurons or other brain cells, inducing the cellular responses in AD that ultimately result in neurodegeneration, and what this means for possible treatments. In the meantime, this model provides a unique possibility to test novel drugs against human microglia for the treatment of AD.
“Overall, this research is an important step toward understanding the mechanisms behind AD. The study provides new insights into the complex ways human microglia respond to AD, which could help researchers develop better treatments for the disease,” concludes Professor De Strooper. “Our findings validate this xenograft model as a powerful tool to investigate the genetics underlying microglial response in Alzheimer's.”
Publication
Xenografted human microglia display diverse transcriptomic states in response to Alzheimer’s disease-related amyloid-β pathology. Mancuso et al. Nature Neuroscience, 2024. DOI: 10.1038/s41593-024-01600-y
This work was funded by the European Research Council, the FWO program, the Methusalem grant, the Belgian Alzheimer Research Foundation (SAO-FRA), the Alzheimer’s Association USA, and the UKDRI-MRC, UK.
India Jane Wise
Joran Lauwers
About the VIB-KU Leuven Center for Brain & Disease Research
Scientists at the VIB-KU Leuven Center for Brain & Disease study how brain cells are organized and how they communicate with each other. These mechanisms reveal and provide insights into what goes wrong in brain diseases such as Alzheimer's, Parkinson's, ALS, and dystonia. This basic work should ultimately lead to new drugs for use against these currently incurable diseases.
About the VIB-UAntwerp Center for Molecular Neurology
Neurodegenerative diseases such as Alzheimer's, epilepsy, and neuromuscular disorders are a major challenge for our society. Research at the VIB-UAntwerp Center for Molecular Neurology begins by studying patients and their family members in order to gain new insights into how these diseases start and develop. Thanks to this knowledge, new therapies can be developed.
.png)