Untangling gut and joint inflammation in arthritis
New study reveals distinct triggers for gut and joint inflammation using germ-free mouse technology.
Ghent, 11 September 2023 – Scientists at the VIB-UGent Center for Inflammation Research shed new light on the relationship between gut and joint inflammation. Their study challenges previous assumptions that joint inflammation is driven by alterations in the gut, including changes in the intestinal microbiome. The new study proves that severe arthritis can develop in the complete absence of intestinal bacteria and intestinal inflammation. Disconnecting this gut-joint axis opens the door to tissue- and patient-specific therapeutic interventions. The work appears in EMBO Molecular Medicine.
The gut-joint axis
Arthritis is a common complication in inflammatory bowel disease (IBD), and vice versa, arthritis patients often develop gut inflammation. This has led to the concept of a 'gut joint axis' – a shared disease pathway that suggests that gut inflammation and microbial triggers can instigate inflammation in the joints
However, correlation does not necessarily equal causation.
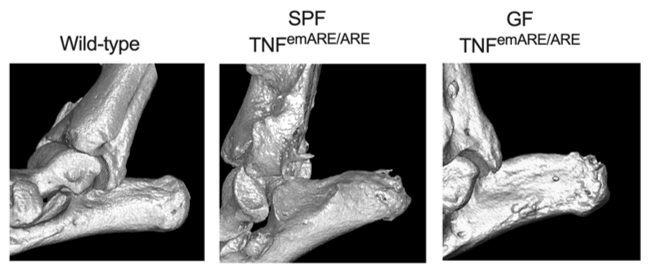
To study the role of the microbiome in various diseases, including arthritis, the lab of Prof. Lars Vereecke (VIB-UGent) established the first Belgian germ-free mouse facility, in which two transgenic mouse models of gut & joint inflammation were established. Surprisingly, when these mice were maintained under sterile conditions, they were protected from intestinal inflammation but still developed severe joint inflammation, indicating that intestinal bacteria drive gut inflammation, but are dispensable for joint inflammation.
"Inflammation in the gut and joints has long been thought to be mechanistically linked, but our study challenges this hypothesis," said Prof. Vereecke. "While shared inflammatory pathways may contribute to both gut and joint inflammation, our research indicates that the triggers to activate these pathways are different in each tissue, and require tissue-specific therapeutic interventions."
Top: germ-free mouse facilities; bottom: arthrosis in ankles of specific pathogen-free (SPF) and germ-free (GF) mice.
Different triggers
These findings indicate that microbial factors can drive gut inflammation, while sterile mechanisms appear to contribute more significantly to joint inflammation. The study's implications are far-reaching, offering potential insights into the development of tissue-specific therapeutic interventions. By understanding the distinct molecular triggers for gut and joint inflammation, researchers may be able to design interventions that target the underlying causes of each condition more precisely.
"Our results illustrate the complexity of inflammatory diseases and emphasize the importance of considering tissue-specific mechanisms," said Alexandra Thiran (VIB-UGent), first author of the study. "Our research opens up new avenues for investigating the drivers of inflammation and developing novel treatments for arthritis and related conditions."

The study not only challenges existing theories about the gut-joint axis but also highlights the need for continued research into the underlying causes of inflammatory diseases. The researchers hope that their findings will inspire further exploration into the intricate connections between different bodily systems and lead to more effective treatments for patients.
Publication
Thiran et al. Sterile triggers drive joint inflammation in TNF and IL-1b dependent mouse arthritis models. EMBO Molecular Medicine, 2023.