Molecular keyhole sheds light on pain and epilepsy
Leuven, 21 April 2026 – Researchers at VIB, VUB, and KU Leuven have identified a tiny binding site, a molecular ’keyhole’, in the TRPM3 ion channel, a crucial sensor in pain signaling. TRPM3 is also linked to rare neurodevelopmental disorders and epilepsy. In a recent study published in Nature Communications, the researchers found that even the slightest change in this keyhole can radically switch the channel’s behavior, explaining how certain mutations can flip the effects of drugs.
“If you have the mirror image of your key or you make a very small change to the key or to the keyhole, suddenly the door might open or close,” says Prof. Thomas Voets, co-lead of the study, and group leader at VIB and KU Leuven.
The study also resulted from a strong collaboration between the center in Leuven and Prof. Janine Brunner from VUB, which also had Dr. Alexander Shkumatov and Dr. Stephan Schenck as co-first authors.
The lock and key model in the TRPM3 channel
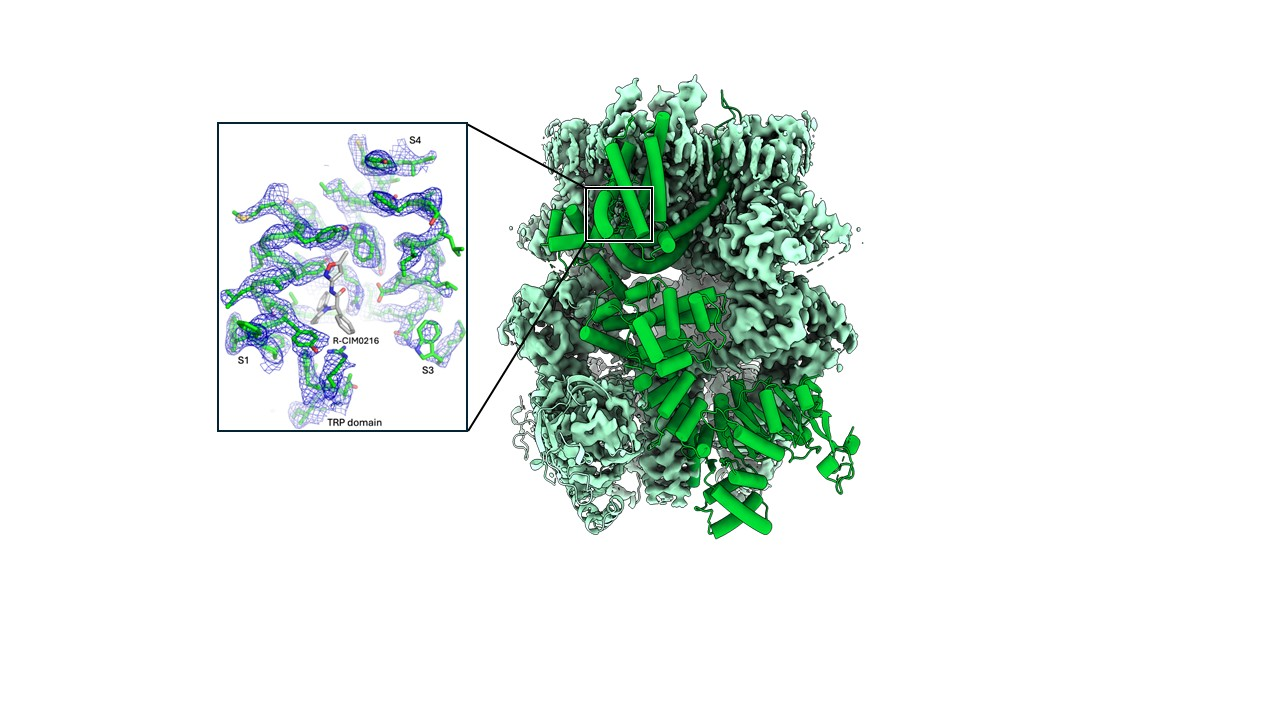
Overactivation of TRPM3 is associated with neurodevelopmental issues and epilepsy. Therefore, therapeutic strategies have been looking into inhibiting this channel, and one of the most common strategies uses isosakuranetin, a plant-derived flavonoid. They observed it has two mirror-image forms: S and R. Surprisingly, only the R form fits the pocket and blocks the channel.
“We discovered that the active form of isosakuranetin is R, not S,” Bahar Bazeli, co-first author of the study. “R is a potent inhibitor of the channel, while S is ineffective.”
The researchers found more. A striking finding is that patient-derived mutations in this pocket can alter drug responses.
“Any small change in this pocket can affect the direction of the effect and also the efficacy,” explains Bazeli. “You can turn an antagonist into an agonist and vice versa. We know now that patients with those specific mutations shouldn’t use the drug everyone else uses. It won’t work for them and would only cause side effects without benefit.”
In other words, R would either become ineffective or trigger the channel instead of inhibiting it. This plasticity has direct clinical implications, as some epilepsy-linked TRPM3 variants lie right in this pocket, which explains why certain medications fail in those patients.

TRPM3 and trigeminal neuralgia: a key to severe pain
Another recent study, published in Cell Reports Medicine, focused on how TRPM3 contributes to one of the most severe pain disorders, trigeminal neuralgia.
Trigeminal neuralgia is caused by injury or compression of the trigeminal nerve in the face . The scientists found that nerve injury and inflammation crank up TRPM3 activity, making facial pain neurons hyperexcitable.
“Trigeminal neuralgia is one of the worst pain syndromes. People call it the suicide disease because it’s so painful ,” notes Prof. Thomas Voets. “We show that inhibiting TRPM3 works surprisingly well in animal models”.
These findings were supported by genetic data: certain TRPM3 gene variants (which likely make the channel more active) are significantly more common in trigeminal neuralgia patients than in the general population. This suggests a genetic predisposition through TRPM3, and linked to the study above, shows why several patients are resistant to general therapies: the different variants cause a different keyhole for the drug, preventing the desired pain-relieving effect.
Master key for targeted therapies
Together, these studies highlight how finding a master key to TRPM3 could be crucial for controlling pain and improving neurological health. By understanding exactly how molecules fit this lock, drug developers can design better ‘keys, which are more potent and selective, to treat TRPM3-related conditions. Researchers are now designing mutant-specific TRPM3 blockers to translate these findings into personalized pain therapies
“Knowing exactly how a molecule fits in this lock helps a lot with developing better and more specific drugs. We are now working on keys that fit even better,” concludes Voets. “These studies open the door to personalized pain medicine, allowing doctors to tailor treatments based on a patient’s TRPM3 ‘keyhole’
Ultimately, unlocking this molecular keyhole could lead to effective new therapies for chronic pain syndromes, neurodevelopmental disorders, and epilepsy.
Publications
Stereoselectivity and functional plasticity of a common ligand-binding pocket in TRPM3. Bazeli, et al. Nature Communications, 2026. DOI:
TRPM3 plays a key role in spontaneous pain and mechanical allodynia in a mouse model of chronic orofacial neuropathy. Desure, et al. Cell Reports Medicine, 2026. DOI: 10.1016/j.xcrm.2026.102645.
Funding
The research was supported by, among others, VIB, KU Leuven, Vrije Universiteit Brussel, Queen Elisabeth Medical Foundation for Neurosciences, and Research Foundation Flanders - FWO.
João Cardoso