New lung cancer model reveals how tumor location shapes the immune response
Brussels, 27 April 2026 – Researchers at VIB and VUB have developed a powerful new way to study how the immune system behaves inside lung tumors. By combining a patient-relevant mouse model with single-cell technologies, the team provides one of the most comprehensive immune maps to date of lung adenocarcinoma, which is the most common subtype of lung cancer. Their work appears in Nature Communications.
“We created a lung cancer model that closely mimics how tumors grow in patients,” says Prof. Damya Laoui (VIB-VUB Center for Inflammation Research). “Combined with a new tracking method for cells, this allow us to tell the difference between immune cells inside the tumor tissue and those that are just passing by in the bloodstream. That distinction makes a big difference. It allows us to see much more clearly how immune cells behave and change once they are inside the tumor.”
A model that better reflects patients
Lung cancer remains the leading cause of cancer-related deaths worldwide, leading to almost 20% of all cancer mortality. Developing effective therapies begins with preclinical studies that mostly rely on subcutaneous tumor models, where cancer cells are implanted under the skin. While practical, these models fail to capture the lung’s unique immune landscape.
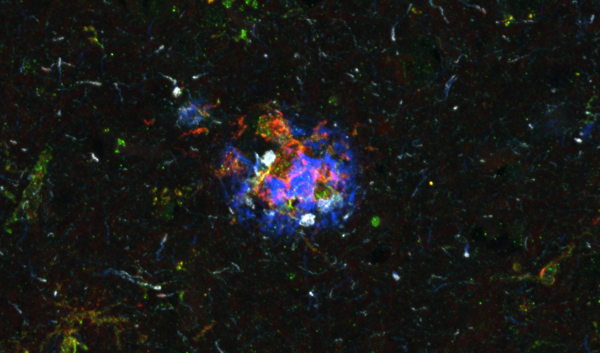
To address this gap, the researchers developed a lung adenocarcinoma model, in which tumors grow directly inside the lung. Crucially, the model allows researchers to dissect tumor nodules separately from adjacent healthy lung tissue, mirroring how patient samples are handled in the clinic. When the team compared their model to human lung adenocarcinoma datasets, they found that it closely reproduced key immune features observed in patients, such as dysfunctional natural killer (NK) cells inside tumors and increased regulatory and exhausted T cells.
“Our goal was to build a model that reflects what we actually see in patients,” says Pauline Bardet (VIB-VUB), PhD student and co-first author of the study. “By placing the tumor in its natural environment, the lung, we capture immune dynamics that are simply absent in subcutaneous models.”

Introducing SEPARATE-Seq
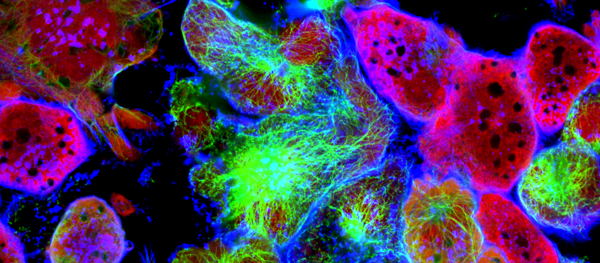
A central innovation of the study is SEPARATE-Seq (Streptavidin Enabled PARtitioning And Tag Evaluation for RNA-Sequencing). In organs like the lung, immune cells are distributed across different compartments: within blood vessels, in the tissue itself, or in the airways. Standard single-cell RNA sequencing cannot easily distinguish between immune cells that are truly infiltrating a tumor and those merely passing through its blood supply. SEPARATE-Seq overcomes this limitation by ‘labeling’ immune cells in the blood.
“Location matters enormously,” explains Prof. Damya Laoui, senior author of the study. “An immune cell inside a blood vessel is not experiencing the same signals as one embedded in tumor tissue. With SEPARATE-Seq, we can finally resolve that difference at single-cell resolution.”
The method is broadly applicable beyond lung cancer and can be used in other diseases where different immune cell populations need to be distinguished.

Immune cells reorganize inside tumors
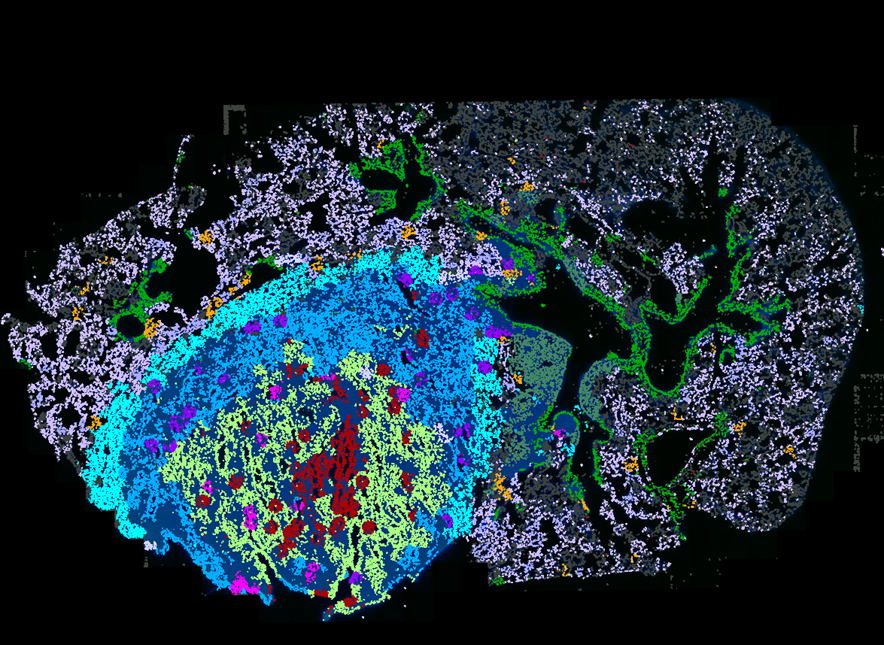
By combining SEPARATE-Seq with spatial transcriptomics, the researchers mapped not only which immune cells are present, but also where they are precisely located inside tumors.
Their analyses revealed several striking spatial patterns. The team identified a ring of lipid-associated tumor-associated macrophages lining the tumor edge, alongside defined hubs of interferon-stimulated immune and non-immune cells within the tumor, that were enriched in specific dendritic cell states. They observed increased infiltration of hypoxic, tumor-associated neutrophils, enrichment of plasma cells in the tumor, and a shift of NK cells toward an immature, dysfunctional state upon tumor entry.
Importantly, many of these features were also observed in human lung adenocarcinoma samples, underscoring the translational relevance of the model.
“This level of spatial and molecular resolution allows us to see how immune cells specialize within defined tumor niches,” says Lize Allonsius, PhD student and co-first author of the study. “It highlights how strongly the tumor microenvironment reshapes immune function.”
A resource for the research community
Beyond establishing the model, the team provides a comprehensive multiomics dataset and makes it accessible through an interactive online tool, offering a valuable resource for researchers studying tumor immunology and lung cancer biology.
“Therapies succeed or fail based on how immune cells behave inside real tumors,” says Laoui. “If our models do not faithfully reflect patient biology, we risk drawing misleading conclusions. With this work, we provide a framework that brings preclinical research one step closer to the clinic.”
Publication
Multiomics immune profiling of a patient-relevant orthotopic lung cancer model using SEPARATE-Seq. Bardet, Allonsius, Hadadi, et al. Nature Communications, 2026.
Funding
This work was supported by Research Foundation Flander — FWO, Kom op tegen Kanker (Stand Up To Cancer Belgium), Stichting tegen Kanker (Foundation Against Cancer), Universitaire Stichting, VIB, and VUB.
Gunnar De Winter