New research brings personalized treatment for Parkinson’s disease a step closer
Leuven, 5 May 2026 – A new study led by researchers from VIB and KU Leuven shows that Parkinson’s disease can be divided into distinct subtypes, helping explain why a single treatment does not work for all patients. Using an machine-learning-driven analysis, the team identified two main groups and five subgroups of the disease, marking an important step toward more personalized therapies. The findings were recently published in Nature Communications.
“We discovered two broad subgroups that can be divided into five smaller groups of parkinsonism”, says Prof. Patrik Verstreken (VIB-KU Leuven Center for Neuroscience)
Parkinson’s disease affects millions of people worldwide and is traditionally defined by its clinical symptoms, including movement difficulties and progressive neurological decline. However, despite being grouped as a single disorder, Parkinson’s can be caused by mutations in many different genes, leading to diverse underlying biological mechanisms. This complexity has challenged the development of effective treatments, as therapies targeting one pathway may not work for all patients.
The new study reveals that these genetically different forms of Parkinson’s can be organized into distinct molecular subtypes, highlighting the need to rethink the disease as a collection of related conditions and opening the door to more targeted therapeutic approaches.
“When clinicians or patients are looking at the disease, they see the clinical symptoms, which unifies people with Parkinson's disease,” says Verstreken. “But when you look under the hood at the molecular level, then you see that they fall into subcategories. And that's important because one drug to target the different molecular dysfunctions in all Parkinson's disease essentially doesn't exist.”
An unbiased analysis to group different Parkinson mutations
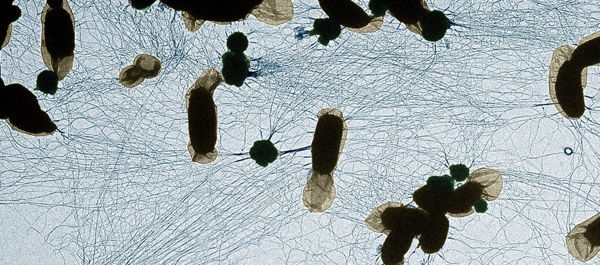
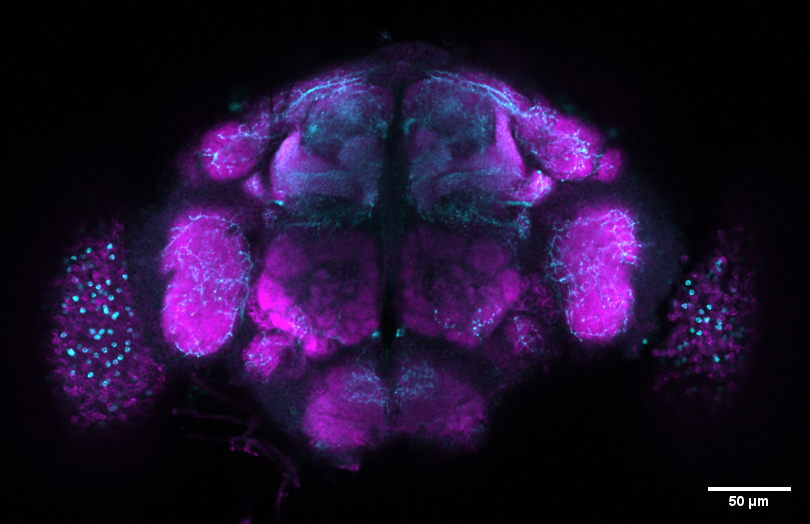
Rather than starting with assumptions about how different genetic mutations could affect the disease, the researchers monitored the behavior of fruit fly models carrying mutations in Parkinson’s-related genes over time and used unbiased computational and machine learning-based methods to identify patterns. By allowing the data to guide the analysis, the team was able to uncover natural groupings of the disease in these animals that would not have been evident using traditional hypothesis-driven methods.

“We came in without any preconceived notions of how a specific mutation would affect our animal model. We took animals with mutations in any of those 24 different genes that are causing the disease, and we just monitored their behavior over periods of time,” adds Dr. Natalie Kaempf, first author of the study.
Together, this unbiased strategy revealed previously hidden structure within Parkinson’s disease, showing that different genetic forms naturally cluster into distinct subtypes.
By moving away from assumptions and letting patterns emerge directly from the data, the study provided a powerful framework for understanding the biological diversity of the disease and guiding future research toward more precise interventions. It also stands as an excellent example of how machine learning can reveal features of disease biology that would otherwise remain impossible to detect, uncovering hidden structure and clinically meaningful variation that are not apparent through conventional approaches.

Potential clinical translation and future perspectives
“We now know that there are different kinds of Parkinson’s disease ,” says Verstreken. “By having these subcategories, we can now go and look within that group of patients with those particular mutations, search specific biomarkers, and develop drugs tailored to each group.”
Researchers managed to cure the Parkinson’s phenotype in animal models by testing compounds in different subgroups. They also observed that different sub-groups respond differently to different compounds.
“When we took a first compound that cured subgroup A and tested it in subgroup B, the latter wasn’t rescued. Our study shows that you can make subgroup-specific drugs that have positive effects and are really specific to that subgroup,” explains Verstreken.
And this unbiased strategy could be adopted into other diseases caused by mutations in multiple genes.
“The same principle can be applied to other types of diseases. Diseases that are caused by mutations in a variety of different genes or environmental factors could be classified according to this principle,” concludes Verstreken.
Publication
Behavioral screening defines the molecular Parkinsonism-related subgroups in Drosophila. Kaempf, et al. Nature Communications, 2026. DOI: 10.1038/s41467-026-70303-8
Funding
This research was supported by, among others, VIB, KU Leuven, the Fund Generet of the King Baudouin Foundation, the European Research Council, and Research Foundation Flanders - FWO. For all funders, see the paper's acknowledgments.
João Cardoso